 An internal audit assessment conducted at a supplier will detail where the supplier meets the ISO 13485 standard and where it does not. Medical device manufacturers increasingly rely on suppliers for specialized materials, software, packaging, and labeling. 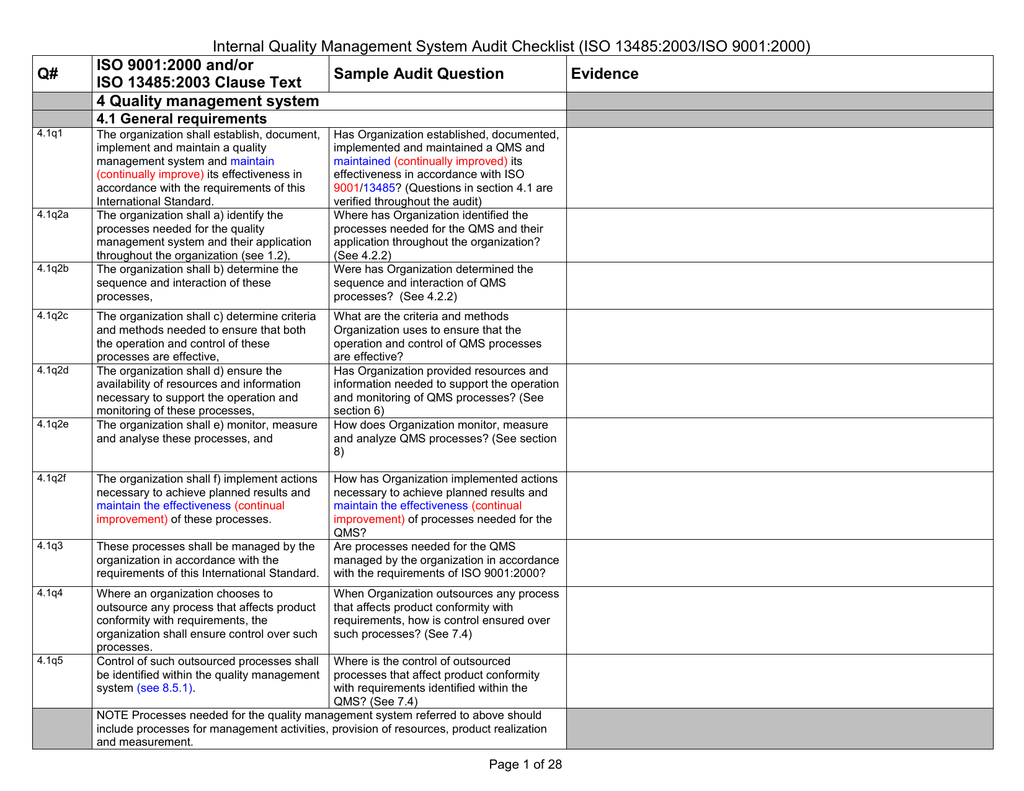
Onboarding and ensuring a potential supplier’s compliance with the standard.All this amounts to a dynamic picture that requires continual vigilance.Ī medical device manufacturer may need an ISO 13485 Internal Audit Assessment when: The ISO 13485 standard is updated periodically. Medical device organizations grow and evolve quickly. When and why is an ISO 13485 Internal Audit Assessment needed? This can include a medical device maker, supplier, external testing organization, and others. It is important to note that the Internal Audit Assessment can be applied to any organization in the medical device supply chain that needs to demonstrate the suitability of its quality management system for customer and regulatory requirements. The checklist may be used as a tool for the audit, and it may be presented, along with an audit summary, as a product of the analysis. 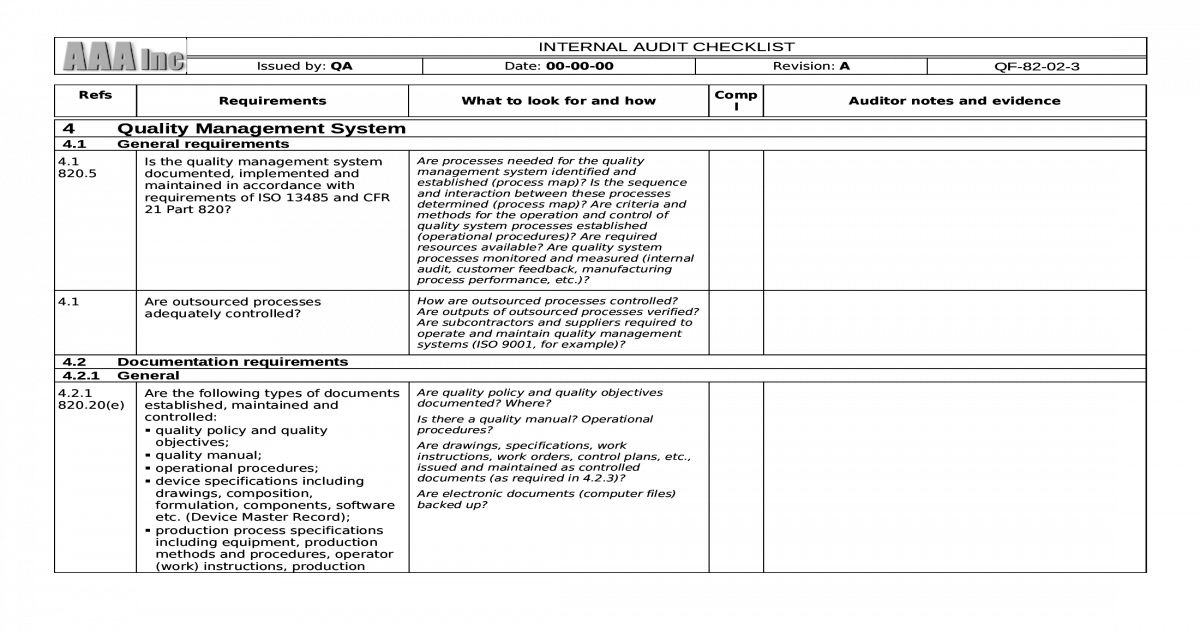
The auditor goes through each requirement of the standard and compares it to the current state in the medical device organization. This is the current revision of the medical device quality management system standard for medical device firms published by the International Organization for Standardization.Įssentially, this analysis can be condensed into a checklist. The analysis will show where the organization is and is not meeting the standard.įor a medical device manufacturer, an ISO 13485 Internal Audit Assessment will systematically compare the current quality management system to the requirements in ISO 13485:2016. Agenda and checklist for a third-party ISO 13485 audit Introduction – ISO 13485 Internal Audit Assessment What is an Internal Audit Assessment?Īn Internal Audit Assessment is a formal, comprehensive comparison of the current state of an organization’s processes and procedures against a standard or regulation. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
